ValedaTM PBM is the first and only FDA-authorized treatment for dry AMD to improve and maintain vision for up to 2 years1*
*The presence of at least 3 medium drusen (>63 μm and ≤125 μm in diameter), or large drusen (>125 μm in diameter), or non-central geographic atrophy.

Valeda™ PBM LIGHTSITE III clinical trial met the predetermined primary efficacy endpoint at Month 212†
84% of Valeda™ PBM–treated patients improved or maintained vision at ~2 years compared to baseline3,4†
More than 60% of Valeda™ PBM–treated patients experienced ≥1-line vision improvement at about 2 years compared to baseline3,4†
Assessing the Efficacy and Safety of Valeda™ PBM
LIGHTSITE III Pivotal Trial Design
Study design
LIGHTSITE III evaluated the efficacy and safety of Valeda™ PBM in a 24-month, double-masked, sham-controlled, parallel design, prospective, randomized, multi-center trial.4,5
Study population
98 patients (145 eyes) aged ≥50 years with ETDRS BCVA letter score between 50 and 75 and a diagnosis of dry AMD defined by the presence of drusen and/or nonfoveal center GA were randomized at a 2:1 ratio into 2 treatment groups.4
Endpoints
Primary efficacy endpoint: Mean BCVA change from baseline to Month 13 or Month 21. Primary safety endpoint: Mean change from baseline to Month 13 or Month 24.4
Valeda™ PBM Improved and Maintained Vision
84% of Valeda™ PBM–treated patients maintained or improved vision at about 2 years compared to baseline3,4†
More than 60% of Valeda™ PBM–treated patients experienced ≥1-line vision improvement at about 2 years compared to baseline3,4†
† n=98 subjects and 145 eyes
No phototoxicity or serious adverse events related
to Valeda™ PBM2,4
Consistent safety in multiple clinical trials


0 reports of phototoxicity in 3
LIGHTSITE trials2

of patients did not report
eye pain4
Non-Invasive Treatment.
Dilation free. Anesthesia free. Speculum free. Injection free.4

Treatment regimen consists of 54 applications administrated in 6 courses over 2 years
¶ 3-5 weeks
‡ For 2 years
Valeda™ PBM Uses the Power of Light
Valeda™ PBM applies 3 science-backed wavelengths to upregulate cellular energy production6,7


590 nm
Increases nitric oxide synthesis and vasodilation which can improve local oxygenation and nutrient delivery6

660 nm
Promotes O2 binding (CuB), stimulates metabolic activity (ATP), and inhibits inflammation and cell death7


850 nm
Drives electron transfer (CuA), stimulates metabolic activity (ATP), and inhibits inflammation and cell death7
Valeda™ PBM’s unique mechanism of action works to restore cellular energy production and improve retinal cellular health5

The retina is rich in mitochondria, and mitochondrial dysfunction is a known cause of vision loss in dry AMD.

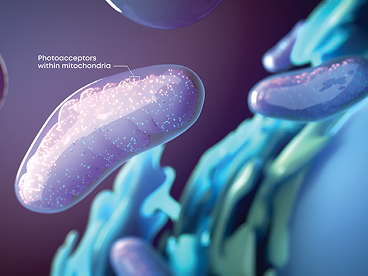
Cytochrome c oxidase (CcO) is a key photoacceptor in the mitochondrial electron transport chain.
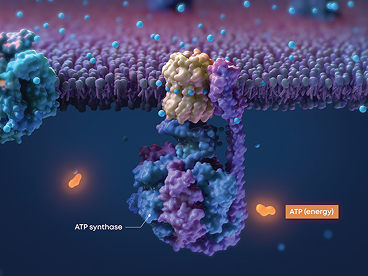

Valeda™ PBM wavelengths activate CcO, enhancing electron transport and mitochondrial adenosine triphosphate (ATP) production, the cell’s major source of energy.
No More Watch and Wait. Treat With Valeda™ PBM
First and only Valeda™ PBM is the first and only FDA-authorized treatment for early and intermediate dry AMD1
Improve and maintain vision
84% of Valeda™ PBM–treated patients improved or maintained vision at ~2 years compared to baseline3,4*
A noninvasive treatment Dilation free, speculum free, anesthesia free, and injection free4
*n=98 subjects and 145 eyes.

Important Product Information:
Indications for Use
The Valeda Light Delivery System is intended to provide improved visual acuity in patients with best-corrected visual acuity of 20/32 through 20/70 and who have dry age-related macular degeneration (AMD) characterized by:
• The presence of at least 3 medium drusen (> 63 μm and = 125 μm in diameter), or large drusen (> 125 μm in diameter), or non-central geographic atrophy, AND
• The absence of neovascular maculopathy or central-involving geographic atrophy.
• After about two years, the Valeda Light Delivery System treatment provides improved mean visual acuity of approximately one line of visual acuity (ETDRS) compared to those not receiving the treatment.
Contraindications for Use
As a precaution, patients have not been tested and should not be treated with Valeda if they have any known photosensitivity to yellow light, red light, or near-infrared radiation (NIR), or if they have a history of light-activated central nervous system disorders (e.g., epilepsy, migraine). In addition, patients should not receive treatment within 30 days of using photosensitizing agents (e.g., topicals, injectables) that are affected by 590, 660, and/or 850 nm light before consulting with their physician.
Precautions
Safety and effectiveness in patient populations and/or conditions excluded from the clinical study has not been established. This includes the following: patients under the age of 50, pregnant or nursing women, current or history of neovascular maculopathy, presence of center involving geographic atrophy (GA) within the central 1mm diameter, media opacities, including cataracts, which might interfere with visual acuity or imaging in the eye, posterior capsule opacification, which might interfere with visual acuity or imaging in the eye, ocular disorder or disease that partially or completely obstructs the pupil, any visually significant disease in any ocular structure apart from dry AMD.
An analysis of the primary effectiveness endpoint (mean BCVA change from baseline for the PBM arm – the mean BCVA change in the Sham arm) showed the following differences between arms for the subgroup of pivotal study patients with early AMD (Beckman Clinical Category Classification):
• At Month 13: +1.90 letters
• At Month 21: -0.10 letters
• At Month 24: +0.29 letters
The eyecare practitioner should consider the observed benefit/risk profile for this sub-population, when contemplating treatment of patients with this classification of Early AMD.
It is possible that treatment benefit may not persist significantly after treatment is stopped. The clinical study provided no significant data concerning the safety and effectiveness of the device should treatments be applied more frequently than described in this manual, or if more than 54 total treatments are delivered per eye.
Twelve (12) eyes (12.9%) in the PBM group and 4 eyes (7.3%) in the Sham group had a fellow eye that had neovascular AMD (nAMD). Of these 5 (41.7%) of 12 eyes in the PBM-treated group converted to nAMD, and 1 (25.0%) of the 4 eyes in the Sham group converted to nAMD. The eye care practitioner should consider the benefit/risk profile in this sub-population and should closely monitor patients whose fellow eye has nAMD.
References:
1. Valeda US Instructions for Use.
2. Valeda Clinical Study Report. 2023. [REF-28073]
3. Valeda US LIGHTSITE III Exhibit Data. [REF-28187]
4. Valeda US User Manual 2025
5. Boyer D, Hu A, Warrow D, et al. 13-Month efficacy and safety evaluation of multiwavelength photobiomodulation in nonexudative (dry) age-related macular degeneration using the Lumithera Valeda Light Delivery System. Retina. 2024;44(3):487-497. doi:10.1097/IAE.0000000000003980
6. Ball KA, Castello PR, Poyton RO. Low intensity light stimulates nitrite-dependent nitric oxide synthesis but not oxygen consumption by cytochrome c oxidase: Implications for phototherapy. J Photochem Photobiol B. 2011;102(3):182-191.
7. Wong-Riley MT, Liang HL, Eells JT, et al. Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: role of cytochrome c oxidase. J Biol Chem. 2005;280(6):4761-4771.Valeda US Instructions for Use.


