When Your Patients Are Free to Get the Most out of Their Day1,2
that’s OPTI-FREE® Freedom

OPTI-FREE® Puremoist® delivers comfort from morning ‘til night2
![]() From the #1 doctor recommended brand of multi purpose solution3
From the #1 doctor recommended brand of multi purpose solution3
![]() Contains HydraGlyde® Moisture Matrix for lasting comfort1
Contains HydraGlyde® Moisture Matrix for lasting comfort1
![]() Dependable, biocompatible POLYQUAD® and ALDOX® dual disinfection system4
Dependable, biocompatible POLYQUAD® and ALDOX® dual disinfection system4

OPTI-FREE® Puremoist® provides superior efficacy against Acanthamoeba5
Of the 4 leading multi-purpose solutions, OPTI-FREE® Puremoist® shows the highest kill against Acanthamoeba trophozoites, which can cause extensive corneal damage and often blindness.5,6
Propidium iodide stains the dead Acanthamoeba cell matter, so the more red, the more dead.
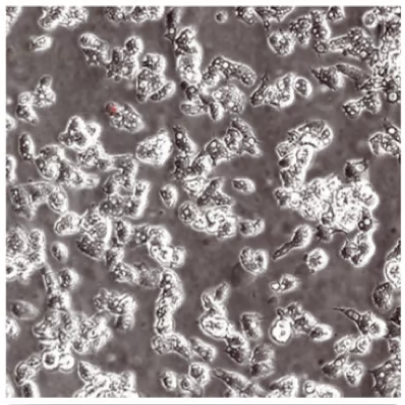

Control – no MPS
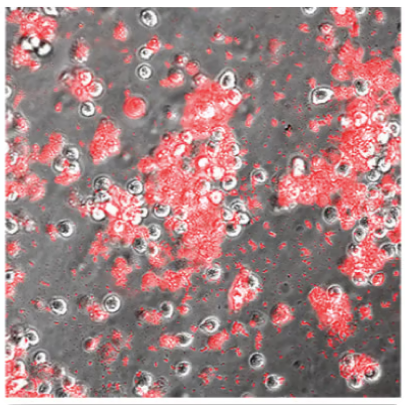

With OPTI-FREE® Puremoist®
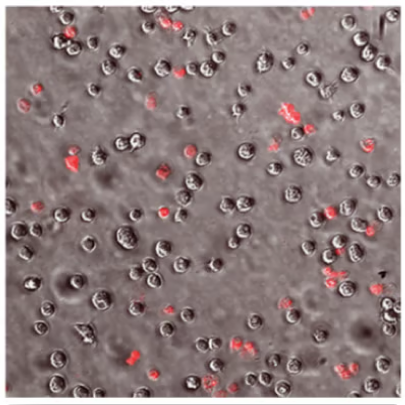

With ReNu Advanced Formula
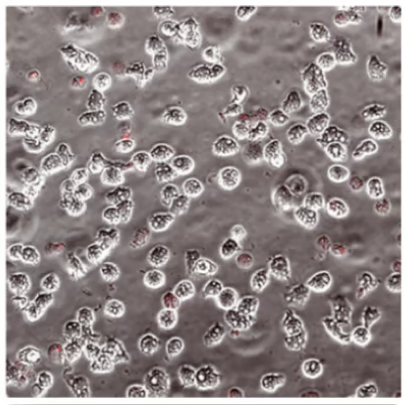

With Biotrue
Evaluation of Acanthamoeba cell death (with propidium iodide) using a stand-alone test. Propidium iodide stains dead Acanthamoeba trophozoite (ATCC 50370) cells red. All images shown at 10x magnification.5
Also available in the OPTI-FREE® family of products
OPTI-FREE® Replenish® offers enhanced comfort, top-notch cleaning, and disinfecting power
![]() Dual disinfectants POLYQUAD® and ALDOX® reduce microorganisms that can cause eye infections
Dual disinfectants POLYQUAD® and ALDOX® reduce microorganisms that can cause eye infections
![]() TearGlyde® reconditioning system creates a shield of comfortable moisture
TearGlyde® reconditioning system creates a shield of comfortable moisture
![]() Removes debris to keep contact lenses clean and clear7
Removes debris to keep contact lenses clean and clear7

REFERENCES
Garofalo R, Lemp J. Clinical trial experience with OPTI-FREE Puremoist MPDS. Contact Lens Spectrum. 2011; 26: Part SPI 44-48
Lally J, Ketelson H, Borajani R, et al. A new lens care solution provides moisture and comfort with today’s CLs. Optician. 2011; 241 (6296): 42-46
Multi-Purpose Solution brands, based on a survey of Eye Care Professionals, Data on File.
Gabriel MM, McAnally C, Bartell J, Walters R, et al. Effect of contact lenses on disinfection efficacy of four multipurpose disinfection solutions. Presented at Association for Research and Vision in Ophthalmology Annual Meeting; May 5-9, 2013; Seattle, WA.
Crary M, Walters R, McAnally C, Gabriel MM, Shannon SP. Differential antimicrobial efficacy of Multi-Purpose Solutions against Acanthamoeba Trophozoites. AAO Poster. 2018.
Awward ST, Petroll WM, McCulley JP, Cavanagh HD. Updates in Acanthamoeba Keratitis. Eye & Contact Lens. 2007; 33: (1): 1-8
OPTI-FREE® Replenish Package Insert. 2019.
IMPORTANT SAFETY INFORMATION
See product instructions for complete wear, care and safety information. ![]()


