Experience Extraordinary
Superior Efficiency for Vitreoretinal and Cataract Surgery*1-4

One unprecedented innovation.
One platform with two configurations.


Alcon’s best-in-class vitreoretinal and cataract surgical capabilities, together in one integrated platform.2-6


Alcon's best-in-class offering for cataract. 2-6
Alcon Experience Academy
For relevant training content from industry thought leaders
FOR THE USE OF REGISTERED MEDICAL PRACTIONERS/HOSPITALS ONLY
UNITY® VCS and CS Important Product Information
Caution: Federal (USA) law restricts this device to sale by, or on the order of, a physician.
INDICATIONS / INTENDED USE:
UNITY VCS:
The UNITY VCS console, when used with compatible devices, is indicated for use during anterior segment (i.e. phacoemulsification and removal of cataracts) and posterior segment (i.e., vitreoretinal) ophthalmic surgery.
In addition, with the optional laser this system is indicated for photocoagulation (i.e., vitreoretinal and macular pathologies), iridotomy and trabeculoplasty procedures.
UNITY CS:
The UNITY CS console, when used with compatible devices, is indicated for use during anterior segment (i.e., phacoemulsification and removal of cataracts) ophthalmic surgery.
Warnings:
Appropriate use of UNITY VCS and CS parameters and accessories is important for successful procedures. The console supports various accessories to perform various surgical procedures.
Accessories include handpieces and probes, as well as tips and sleeves when necessary. Different accessories are required for different procedures and operating modes.
Test for adequate irrigation and aspiration flow, reflux, and operation of each accessory prior to entering the eye.
The consumables used in conjunction with ALCON® instrument products constitute a complete surgical system. To avoid the risk of a patient hazard, do not mismatch consumable
components or use settings not specifically adjusted for particular consumable component combinations.
AEs/Complications:
Inadvertent activation of functions that are intended for priming or tuning accessories while the accessory is in the eye can create a hazardous situation that could result in patient injury.
During any ultrasonic procedure, metal particles may result from inadvertent touching of the ultrasonic tip with a second instrument. Another potential source of metal particles resulting
from any ultrasonic handpiece may be the result of ultrasonic energy causing micro abrasion of the ultrasonic tip.
ATTENTION: Refer to the Directions for Use for the accessories/consumables and User Manual for a complete listing of indications, warnings, cautions and notes.
References:
1. Alcon Data on File, 2024. [REF-24644].
2. Alcon Data on File, 2024. [REF-24576].
3. Alcon Data on File, 2024. [REF-24615].
4. Alcon Data on File, 2024. [REF-24379].
5. UNITY® Phaco Handpiece Directions for Use.
6. UNITY® VCS and CS User Manual, 2024.
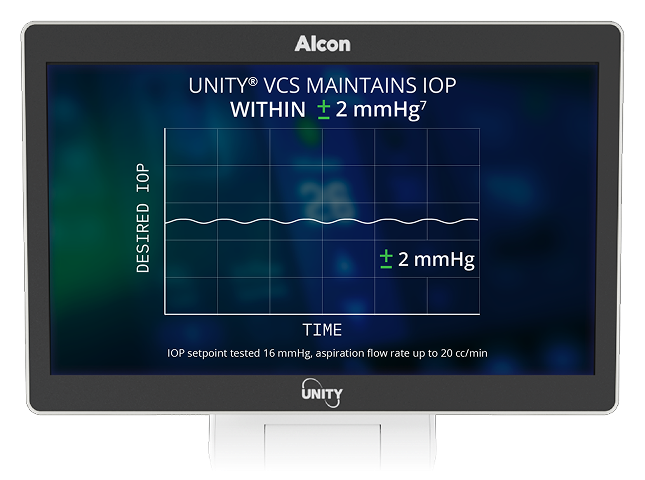
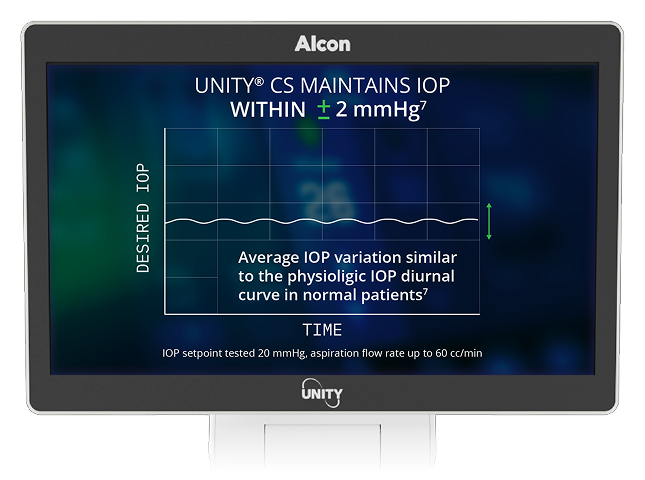
7. Alcon Data on File, 2024. [REF-24899].
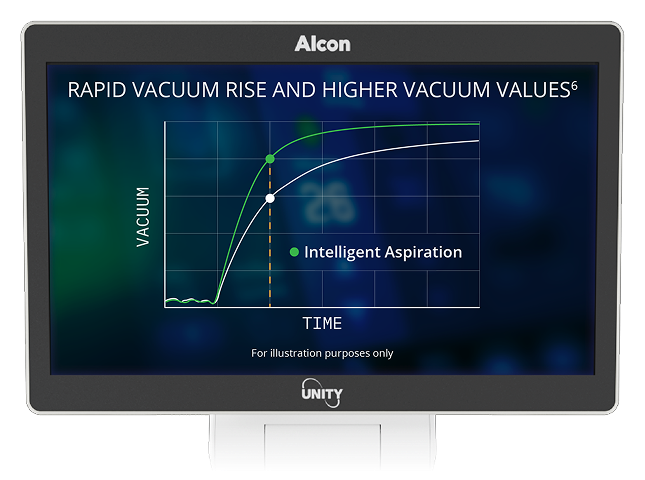
8. Alcon Data on File, 2024. [REF-24760].
9. Alcon Data on File, 2024. [REF-25562].
10. Suzuki H, Igarashi T, Takahashi H. Effect of a new phacoemulsification and aspiration handpiece on anterior chamber stability. J Cataract Refract Surg. 2023;49(1):91-96.
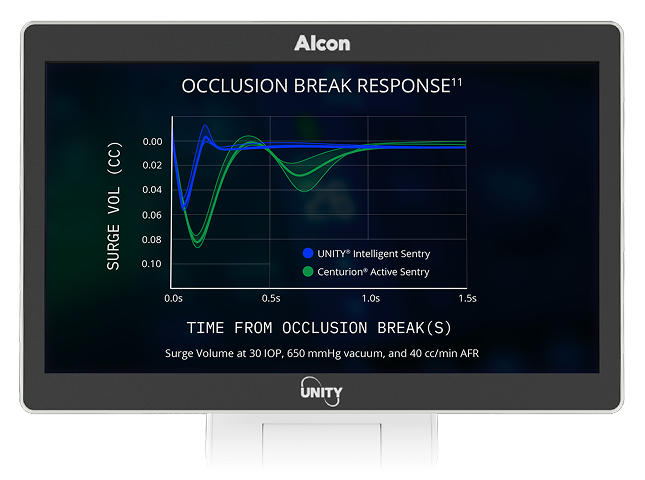
11. Alcon Data on File, 2019. [REF-26073].
12. Irannejad A, Tambat S, Abulon DJK. Retropulsion and Mass flow of 27-gauge Vitrectomy Probes: Comparison of Dual-blade/flattipped probes and single-blade/beveled probes. 2021. Poster presentation.
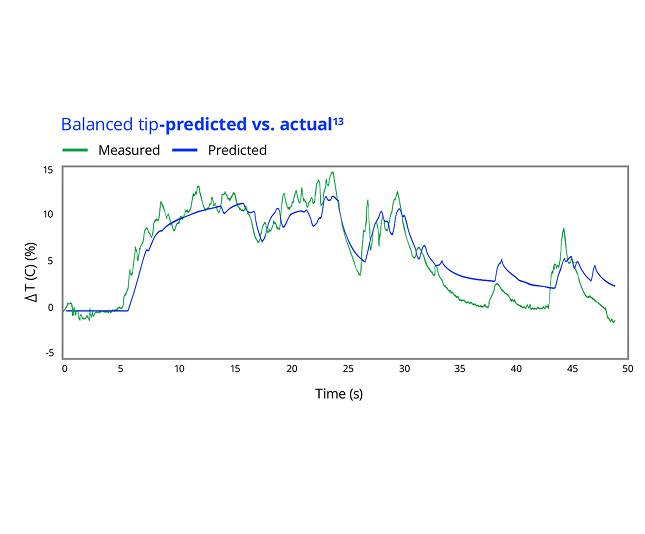
13. Alcon Data on File, 2025. [REF-26793].
14. Alcon Data on File, 2024. [REF-25374].
15. Ayres BD, Gupta OP, Davis JS, et al. Economic impact analysis of custom pak on cataract and vitreoretinal surgery in the United States. Clinic Economics and Outcomes Research. 2022;31:715-30.