AIR OPTIX™ NIGHT & DAY™ AQUA
A flexible lens-wearing experience designed for up to 30 nights of continuous wear1†
†Extended wear for up to 30 continuous nights, as prescribed by an eye care professional.

Busy patients need clear and comfortable vision,
whether it’s in the middle of the night…
…or first thing in the morning
AIR OPTIX™ NIGHT & DAY™ AQUA can help these patients with SmartShield® Technology
AIR OPTIX™ NIGHT & DAY™ AQUA contact lenses are intended for up to 30 days and nights of continuous wear.†
*Dk/t = 175 @ -3.00D. Other factors may impact eye health.
†Extended wear for up to 30 continuous nights, as prescribed by eye care professional
AIR OPTIX™ NIGHT & DAY™ AQUA Offers Overall Deposit Resistance And Wettability That Outperforms Other Lenses
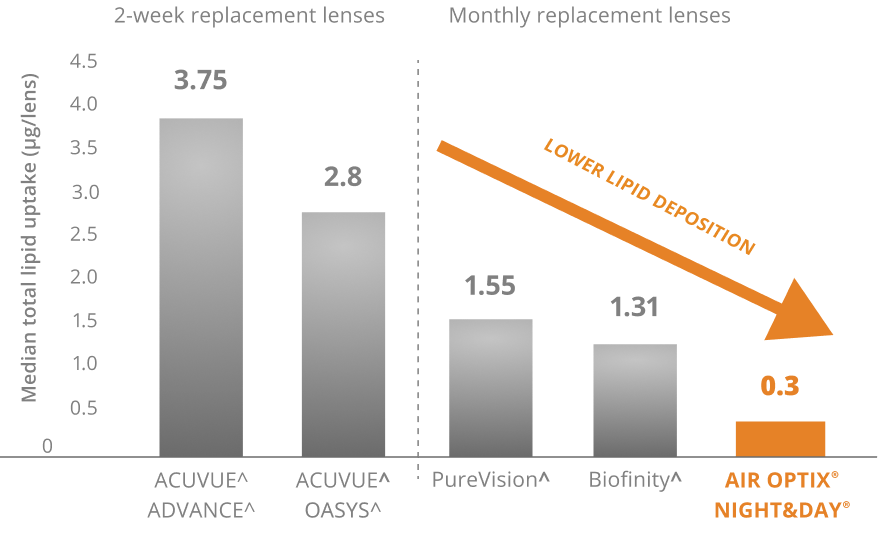
Excellent Lipid Deposit Resistance3,4
Lenses worn daily through manufacturer-recommended replacement period. AOSEPT PLUS® Cleaning & Disinfecting Solution used for cleaning and disinfection. Median values represented.
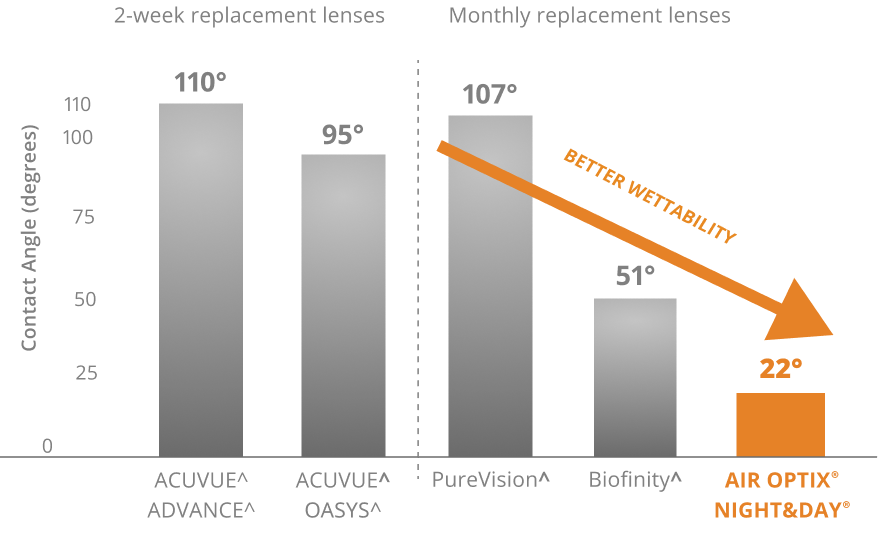
Excellent Wettability2†
In vitro measurement of unworn lenses, median values represented, n=20.
*All differences between AIR OPTIX™ NIGHT & DAY™ AQUA contact lenses and competitive brands statistically significant (p≤0.0001).
Compared to Biofinity, PureVision, ACUVUE OASYS, ACUVUE ADVANCE contact lenses. Comparator products may not be available in all markets.
^Trademarks are the property of their respective owners
†Based on median out-of-package contact angle measurements as measured by the static sessile drop technique
A very breathable lens5
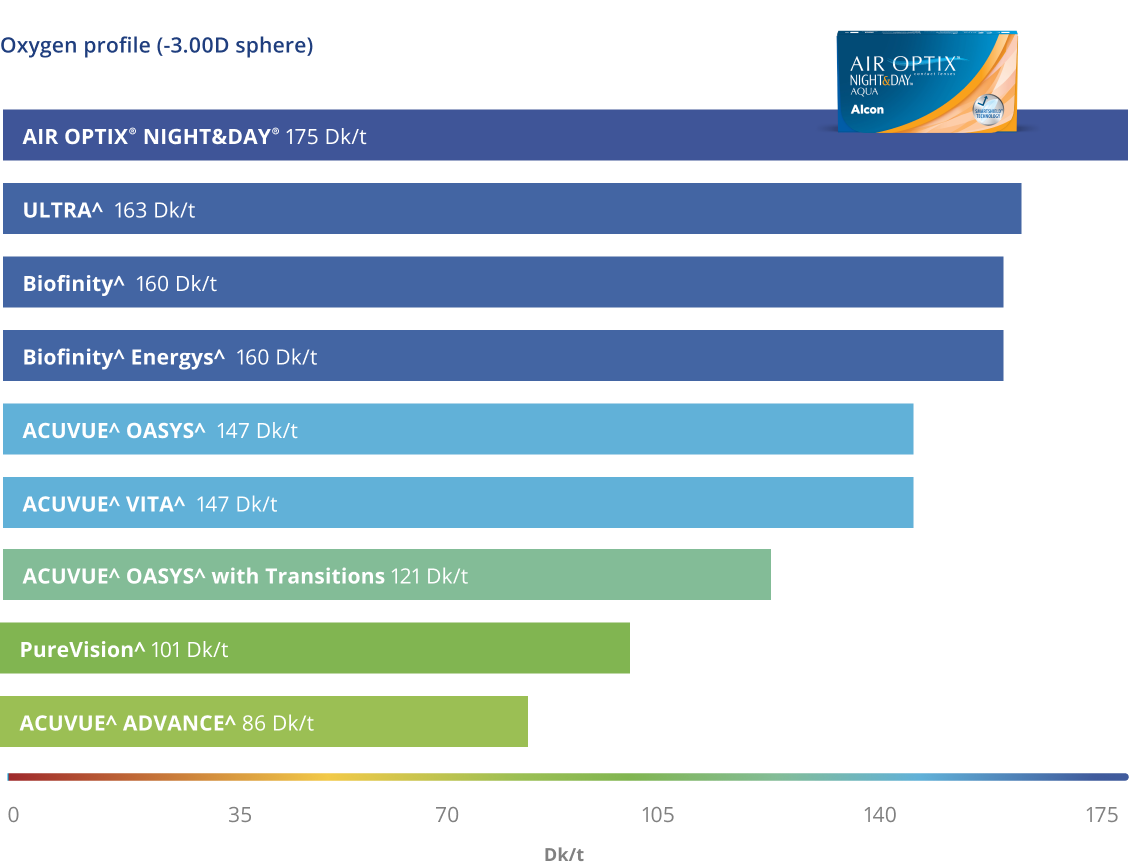
ǂIn an ex vivo assessment compared to ACUVUE^ ADVANCE^, ACUVUE^ OASYS^, PureVision^, Biofinity^ (n=20 for all contact lenses; p<0.05)
^Trademarks are the property of their respective owners.
Comparator products may not be available in all markets.
AIR OPTIX™ NIGHT & DAY™ AQUA has a low rate of microbial keratitis (MK)6
A rigorous post-market surveillance study of nearly 5,000 lotrafilcon A contact lens wearers for one year, practices estimated the overall annual rate was:
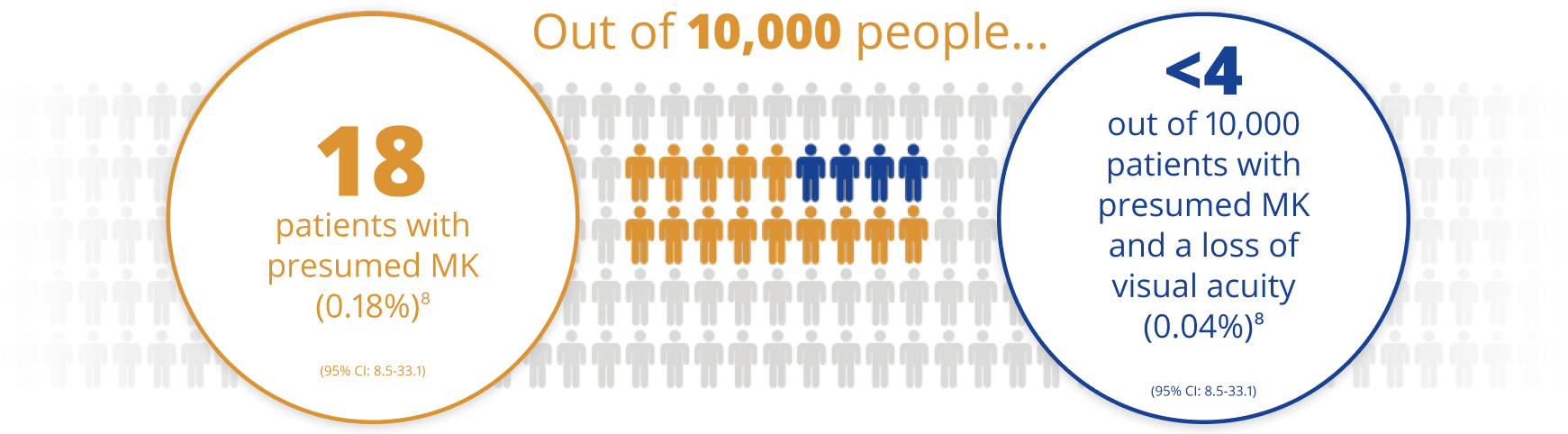
For those who were able to continuously wear the lenses 3-4 weeks, there was a lower rate of presumed MK than for those with <3 weeks of wear (p=0.02).
*Based on calculated overall annual rate of MK
To help your patients get the most from their contact lenses, offer AOSEPT PLUS® and OPTI-FREE® PureMoist®

AIR OPTIX™ NIGHT & DAY™ AQUA contact lenses technical specification



AIR OPTIX™ plus HydraGlyde™ Multifocal
A multifocal contact lens designed to provide outstanding comfort from day 1 to day 30.7-9,10*


AIR OPTIX™ plus HydraGlyde™ for Astigmatism
Outstanding stability with a predictable fit, and outstanding
comfort from day 1 to day 30 for your astigmatic patients.7-9,11*
*Based on a clinical study with AIR OPTIX® AQUA AIR OPTIX® for Astigmatism and AIR OPTIX® AQUA MULTIFOCAL Contact lenses. n = 117.
Reference Links
-
Bergenske P et al. Long term clinical results: 3 years of up to 30 nights continuous wear of lotrafilcon A and daily wear of low-Dk/t hydroge lenses. Eye Contact Lens 2007;33(2):74-80.
-
Alcon data on file, 2005. R-002-TBI-45054-03-B Advancing Contact Angle of Silicone Hydrogel Lenses. Available on request.
-
Nash W, Gabriel M, Mowrey-McKee M. A comparison of various silicone hydrogel lenses; lipid and protein deposition as a result of daily wear. Optom Vis Sci. 2010;87: E-abstract 105110.
-
Nash WL, Gabriel MM. Ex vivo analysis of cholesterol deposition for commercially available silicone hydrogel contact lenses using a fluorometric enzymatic assay. Eye Contact Lens. 2014;40(5):277-282.
-
Based on published manufacturer-provided DK/T values in Tylers Quarterly, 2019
-
Schein O, McNally J, Katz J, Chalmers R. The incidence of microbial keratitis among wearers of a 30-day silicone hydrogel extended-wear contact lens. Ophthalmology. 2005;112(12):2172-79.
-
Eiden SB, Davis R, Bergenske P. Prospective study of lotrafilcon B lenses comparing 2 vrsus 4 weeks of wear for objective and subjective measures of health, comfort, and vision. Eye Contact Lens. 2013;39(4):290-294.
-
Lemp J, Kern J. A comparison of real time and recall comfort assessments. Optom Vis Sci. 2016;93:E-abstract 165256.
-
Based on a 30-day clinical study of 75 habitual lotrafilcon B lens wearers; Alcon data on file, 2017.
-
Alcon data on file, 2016. Alcon Multifocal Precision Profile Contact Lens Design. Available on request.
-
Alcon data on file, 2005. P-242-C-007 1, 2 & 3 O2 OPTIX Toric DW US Comparative Trials. Available on request
-
Alcon data on file, 2015. In vitro biological deposition comparison of Air Optix. Available on request
ALWAYS READ THE LABEL AND FOLLOW THE DIRECTIONS FOR USE.
All content on this website is for informational purposes only, always talk to your health professional regarding your eye health or medical conditions.







