AIR OPTIX® NIGHT & DAY® AQUA
Een flexibele lenservaring, ontwikkeld om tot 30 nachten continu te worden dragen†2

Klanten met een druk leven hebben behoefte aan helder en comfortabel zicht, of het nu midden in de nacht is...
... of 's morgens vroeg
AIR OPTIX® NIGHT & DAY® AQUA® kan deze klanten helpen dankzij de SmartShield®-technologie en het ultieme ademend vermogen.3
AIR OPTIX® NIGHT & DAY® AQUA® contactlenzen zijn goedgekeurd door de FDA (de Amerikaanse toezichthouder op voeding en geneesmiddelen) om tot 30 dagen en nachten continu te worden gedragen.
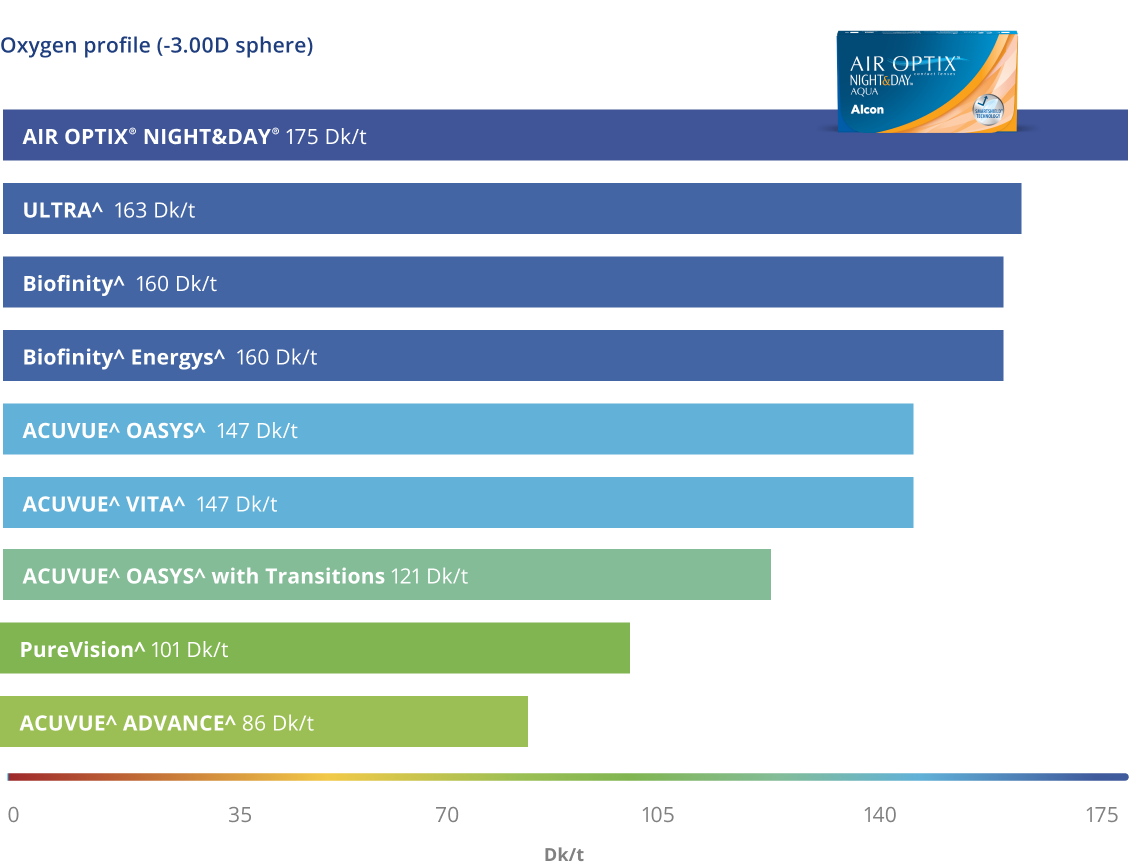
* Dk / t = 175 @ -3.00D. Andere factoren kunnen de gezondheid van het oog beïnvloeden.
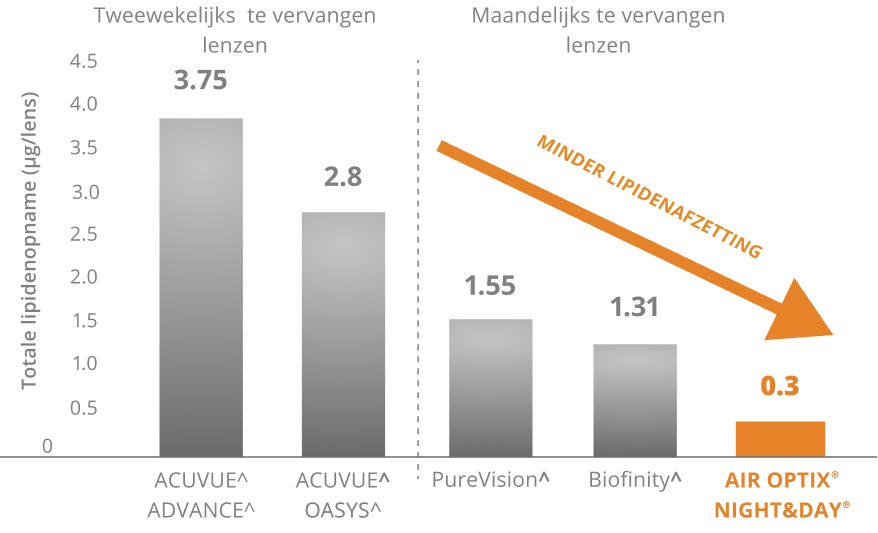
AIR OPTIX® NIGHT & DAY AQUA® biedt een uitzonderlijke weerstand tegen afzettingen en heeft een bevochtigbaarheid die andere lenzen overtreft
Superieure weerstand tegen lipidenafzetting4,5
De lenzen werden dagelijks gedragen tijdens de door de fabrikant aanbevolen vervangingsperiode.
Het meest ademende lensmateriaal op de markt6*
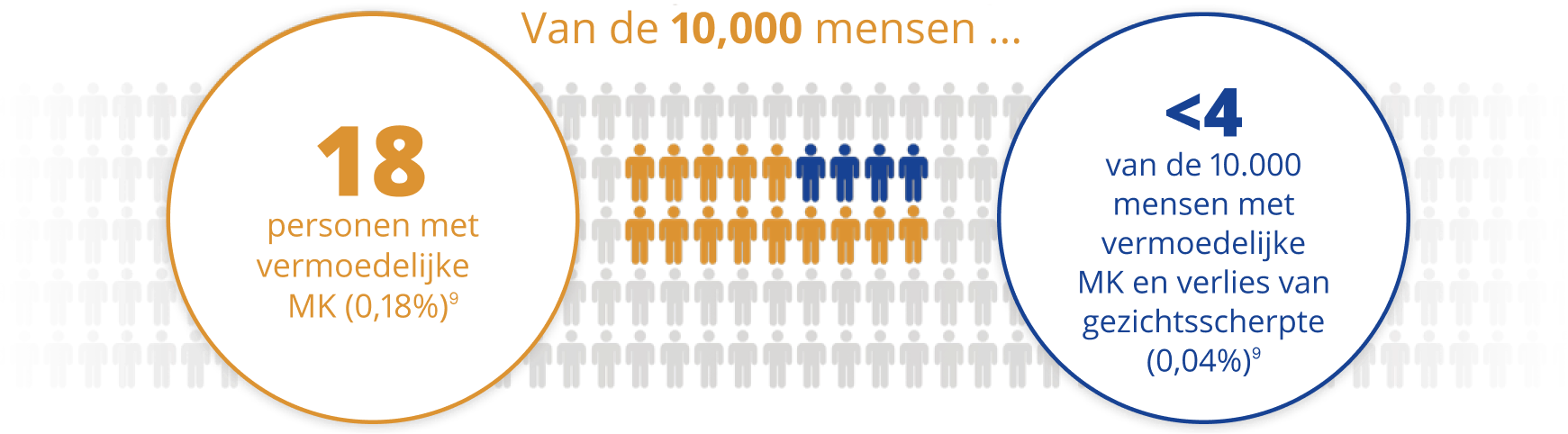
AIR OPTIX® NIGHT & DAY AQUA® heeft een laag percentage microbiële keratitis (MK)7*
Een streng Post Market Surveillance-onderzoek onder bijna 5 000 lotrafilcon A-contactlensdragers gedurende een jaar, en in meer dan 100 klinische praktijken gaf volgende resultaten:
Bij degenen die de lenzen ≥3 weken continu konden dragen, was er een lager percentage vermoedelijke MK dan bij degenen die de lenzen <3 weken droegen (p = 0,02).
* Gebaseerd op het berekende totale jaarlijkse percentage van MK
Productinformatie over AIR OPTIX® NIGHT & DAY® AQUA® contactlenzen



AIR OPTIX® plus HydraGlyde® Multifocal
De multifocale contactlens voor uitstekend comfort, van dag 1 tot dag 30.1-3*


AIR OPTIX® plus HydraGlyde® for Astigmatism
Uitstekende stabiliteit met een voorspelbare aanpassing. Uitstekend comfort voor uw astigmatische klanten van dag 1 tot dag 30.1-3,20
*Gebaseerd op klinische studies met AIR OPTIX® AQUA-contactlenzen.
Referenties
^Trademarks are the property of their respective owners.
†Extended wear for up to 30 continuous nights, as prescribed by an eye care professional.
1. AIR OPTIXTM NIGHT & DAYTM AQUA package insert
2. Based on the ratio of lens oxygen transmissibilities; Alcon data on file, 2009, 2010.
3. Lemp J, Kern J. On-eye performance of lotrafilcon B lenses packaged with a substantie wetting agent. Poster presented at Optometry's Meeting, the Annual Meeting of the American Optometric Association; June 21-25 2017;: Washington D.C.
4. Nash W, Gabriel M, Mowrey-McKee M. A Comparison of various silicone hydrogel lenses; lipid and protein deposition as a result of daily wear. Optom Vis Sci.2010;87; E-abstract 105110.
5. Nash WL, Gabriel MM. Ex vivo analysis of cholesterol deposition for commercially available silicone hydrogel contact lenses using a fluorometric enzymatic assay. Eye Contact Lens. 2014;40(5):227-282.
6. Based on published manufacturer-provided DK/T values in Tylers Quarterly, 2021.
7. Schein O, McNally J, Katz J, Chalmrs R. The incidence of microbial keratitis among wearers of a 30-day silicone hydrogel extended-wear contact lens. Ophtalmology. 2005;112(12):277-282.
8. Eiden SB, Davis R, Bergenske P. Prospective study of lotrafilcon B lenses comparing 2 versus 4 weeks of wear for objective and subjective measures of health, comfort and vision. Eye Contact Lens. 2013;39(4):290-294.
9. Lemp J, Kern J. A comparison of real time and recall comfort assessments. Optom Vis Sci. 2016;93 E-abstract 165256.
10. Based on a 30-day clinical study of 75 habitual lotrafilcon B lens wearers; Alcon data on file, 2017.
11. In a randomized, subject-masked, multi-site clinical study with over 150 patients; significance demonstrated at the 0.05 level. Alcon data on file, 2005.
Voor indicaties, contra-indicaties en waarschuwingen verwijzen wij u naar de gebruiksaanwijzing van het relevante product. Contactlenzen, contactlensvloeistoffen en producten voor droge ogen (m.u.v. Systane® LID WIPES) zijn medische hulpmiddelen.







