Valeda™ PBM is the first FDA-authorized and CE marked treatment for dry AMD to improve and maintain vision for up to 2 years1,8*
*Dry AMD eyes with: 3 medium drusen, or 1 large drusen, or non-central involving GA; and with BCVA between 20/32 - 20/70. (i.e., early and intermediate dry AMD, and a sub-set of late dry AMD).

† n=98 subjects and 145 eyes
Assessing the Efficacy and Safety of Valeda™ PBM
LIGHTSITE III Pivotal Trial Design
Valeda™ PBM Improved and Maintained Vision
84% of Valeda™ PBM–treated patients maintained or improved vision at about 2 years compared to baseline.3,4†
More than 60% of Valeda™ PBM–treated patients experienced ≥1‑line vision improvement at about 2 years compared to baseline.3,4†
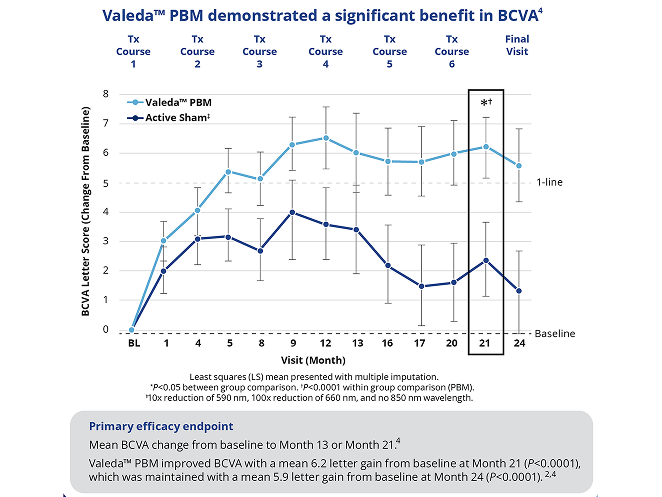
† n=98 subjects and 145 eyes
No phototoxicity or serious adverse events related
to Valeda™ PBM2,4
Consistent safety in multiple clinical trials
Non-Invasive Treatment.
Dilation free. Anesthesia free. Speculum free. Injection free.4

Treatment regimen consists of 54 applications administrated in 6 courses over 2 years
¶ 3-5 weeks
‡ For 2 years
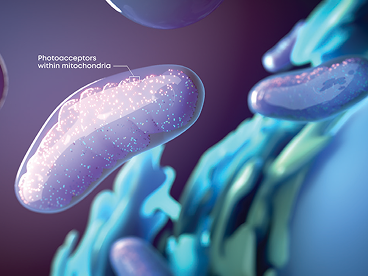
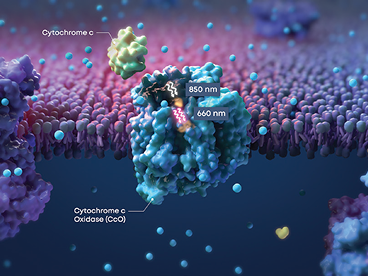
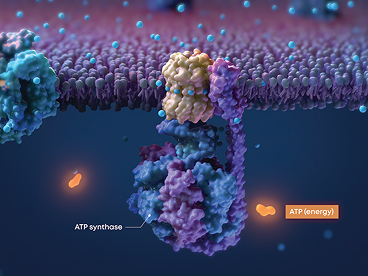
Valeda™ PBM Uses the Power of Light
Valeda™ PBM applies 3 science-backed wavelengths to upregulate cellular energy production6,7
Valeda™ PBM’s unique mechanism of action works to restore cellular energy production and improve retinal cellular health5
No More Watch and Wait. Treat With Valeda™ PBM
First and only Valeda™ PBM is the first FDA‑authorized and CE‑marked treatment for dry AMD to improve and maintain vision for up to two years.1,8*
Improve and maintain vision
84% of Valeda™ PBM–treated patients improved or maintained vision at ~2 years compared to baseline.3,4*
A noninvasive treatment Dilation‑free, speculum‑free, anesthesia‑free, and injection‑free.4
*n=98 subjects and 145 eyes.

Alcon medical device(s) comply with the current legislation for medical devices. Please refer to relevant product instructions for use for complete list of indications, contraindications and warnings.
References:
1. Valeda US Instructions for Use.
2. Valeda Clinical Study Report. 2023. [REF-28073]
3. Valeda US LIGHTSITE III Exhibit Data. [REF-28187]
4. Valeda US User Manual 2025
5. Boyer D, Hu A, Warrow D, et al. 13-Month efficacy and safety evaluation of multiwavelength photobiomodulation in nonexudative (dry) age-related macular degeneration using the Lumithera Valeda Light Delivery System. Retina. 2024;44(3):487-497. doi:10.1097/IAE.0000000000003980
6. Ball KA, Castello PR, Poyton RO. Low intensity light stimulates nitrite-dependent nitric oxide synthesis but not oxygen consumption by cytochrome c oxidase: Implications for phototherapy. J Photochem Photobiol B. 2011;102(3):182-191.
7. Wong-Riley MT, Liang HL, Eells JT, et al. Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: role of cytochrome c oxidase. J Biol Chem. 2005;280(6):4761-4771.Valeda US Instructions for Use.
8. Alcon Data on File, 2025. REF-27576